Predicting EPR¶
Here we will demonstrate the basics on how to predict EPR properties and spectra using ORCA, that can even include many of the complications involved such as spin-orbit coupling and other relativistic effects.
The CH3 radical¶
The methyl radical is a planar molecule, and its planarity was in part confirmed by EPR spectroscopy. It has a \(g\) of \(2.0025\) [Gordy1966] and hyperfine couplings (HFC) \(A_{xx} = -31.4, A_{yy} = -93.7 ,\) and \(A_{zz} = -60.30\) MHz [Dmitriev2004], let's try to predict that from first principles!
First, the geometry can be optimized using:
!B3LYP DEF2-TZVP OPT
* xyz 0 2
C 3.20276693914010 2.91777006829308 -1.46614904250634
H 4.21682401439677 3.22254500904168 -1.25560882817865
H 2.69109288861722 3.31313693290047 -2.33037576806431
H 2.69057615784592 2.24676798976475 -0.79431636125068
*
and you get the trigonal planar coordinates, as expected:
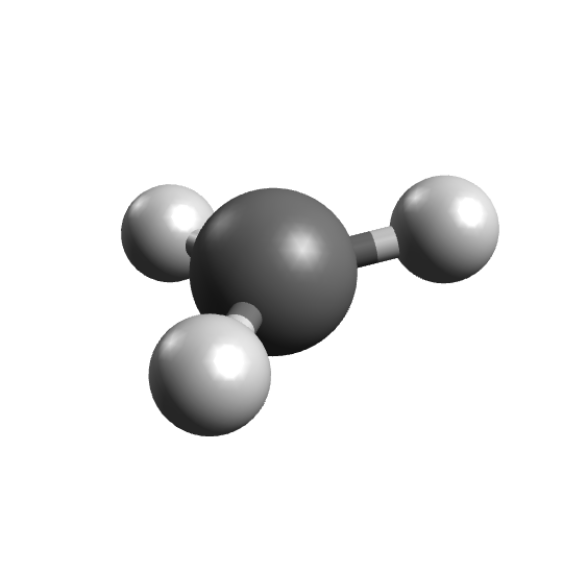
The next step is then to calculate the EPR properties, and that can be done by setting the %EPRNMR options. The important point here is that these are the only options on the ORCA input that should come after the geometry section, and never before.
An input to compute the g tensor and HFCs for the hydrogen atoms, using the B3LYP functional could be:
!B3LYP EPR-II AUTOAUX
* XYZFILE 0 2 CH3_opt.xyz
%EPRNMR
GTENSOR TRUE
NUCLEI = ALL H {AISO, ADIP, AORB}
END
and it should be given with some comments:
The GTENSOR flag must be set to TRUE in order to compute it, the default is FALSE.
The NUCLEI defines the atoms for the HFC calculation. ALL H calculates the HFC on all hydrogens, or use ALL N, ALL C and so on for different atoms. You can also use NUCLEI = 1,5,8, to give one list per atom type with the atom numbering staring from 1.
The AISO, ADIP and AORB flags request the isotropic part of the HFC, its dipolar part and 2nd order contribution from SOC respectively.
The AORB term is costly, but contributes very little to organic molecules without heavy atoms and could be neglected in those cases.
A large basis is recommended for EPR properties. Here we used the special basis EPR-II, for more info one can check the manual available from the ORCA forum.
The spin-orbit coupling integrals are calculated using the SOMF(1X) [Neese2005] method by default, but more efficient options are also available.
Warning
If you are using a list of atoms, like NUCLEI = 1,5,8, there should be one list for each atom type!
The output file¶
After a successful single point calculation, the first step is the computation of the integrals used for the SOC treatment:
------------------------------------------------------------------------------
ORCA SPIN-ORBIT COUPLING CALCULATION
------------------------------------------------------------------------------
GBW file ... epr.gbw
Input density ... epr.scfp
Output integrals ... epr
Operator type ... Mean-field/Effective potential
One-Electron Terms ... 1
Coulomb Contribution ... 2
Exchange Contribution ... 3
Correlation Contribution ... 0
Maximum number of centers ... 4
and then comes the:
------------------------------------------------------------------------------
ORCA EPR/NMR CALCULATION
------------------------------------------------------------------------------
GBWName ... epr.gbw
Electron density file ... epr.scfp
Spin density file ... epr.scfr
Spin-orbit integrals ... epr
Origin for angular momentum ... Center of electronic charge
Coordinates of the origin ... 6.05064281 5.51881078 -2.76743570 (bohrs)
Please notice that the "Origin for angular momentum" here is "Center of electronic charge", and that means these results are not gauge-invariant and can change depending on your coordinates.
Usually, with a sufficiently large basis, the results should be almost invariant, but if one wants the exact results, gauge-invariant atomic orbitals (GIAOs) are needed [Ditchfield1973], [Pulay1990]. These can be constructed from all basis sets, and to use them, just add ORI GIAO to the %EPRNMR section:
%EPRNMR
GTENSOR TRUE
NUCLEI = ALL H {AISO, ADIP, AORB}
ORI GIAO
END
Note
The calculation of the integrals involving GIAOs is much more costly than the regular ones, so be prepared for longer calculation times.
g Tensor¶
If the following CP-SCF converges, you will get the g tensor matrix:
-------------------
ELECTRONIC G-MATRIX
-------------------
The g-matrix:
2.0027537 0.0001534 0.0001065
0.0001534 2.0024625 -0.0002479
0.0001065 -0.0002479 2.0026476
gel 2.0023193 2.0023193 2.0023193
gRMC -0.0001271 -0.0001271 -0.0001271
gDSO(tot) 0.0000289 0.0000680 0.0000680
gPSO(tot) 0.0000034 0.0005594 0.0005595
---------- ---------- ----------
g(tot) 2.0022245 2.0028196 2.0028196 iso= 2.0026213
Delta-g -0.0000948 0.0005003 0.0005004 iso= 0.0003020
Here the components of the g tensor are specified (see [Neese2001]) and the total components for the x, y and z directions are printed after g(tot), with the isotropic value afterwards. The Delta-g shown is the difference from the free-electron g value.
As you can see, the predicted value is already quite close to the experimental value, considering we completely ignored any matrix effects and its largest deviation from the free-electron comes due to SOC effects, as suggested by Gordy [Gordy1966].
Hyperfine couplings¶
After the g tensor calculation, comes the HFC for each atom:
-----------------------------------------
ELECTRIC AND MAGNETIC HYPERFINE STRUCTURE
-----------------------------------------
-----------------------------------------------------------
Nucleus 1H : A:ISTP= 1 I= 0.5 P=533.5514 MHz/au**3
Q:ISTP= 2 I= 1.0 Q= 0.0029 barn
-----------------------------------------------------------
Solving for nuclear perturbation:
Forming RHS of the CP-SCF equations ...
Here "I=" indicates the spin of that given atom and "P=" is the proportionality factor \(\mu_e \mu_N \beta_e \beta_N\) used. After that, another CP-SCF solution is requested and the HFC information is printed:
Raw HFC matrix (all values in MHz):
-----------------------------------
-30.0552 10.8247 7.4998
10.8247 -77.0049 20.6090
7.4998 20.6090 -92.4037
A(FC) -66.5005 -66.5005 -66.5005
A(SD) 41.0835 -0.8524 -40.2311
A(ORB+DIA) 0.0114 -0.0006 0.0271 A(PC) = 0.0126
A(ORB) 0.0065 0.0006 0.0258 A(PC) = 0.0109
A(DIA) 0.0050 -0.0012 0.0013 A(PC) = 0.0017
---------- ---------- ----------
A(Tot) -25.4056 -67.3536 -106.7045 A(iso)= -66.4879
Again, the individual components are printed and the A(iso) is shown in the end. As you can see, the results are in quite good agreement with the experimental data.
At the very end, the ORCA EULER output is printed, with information about the relative angle between the g-tensor and the HFCs, which have an arbitrary relationship from the calculation:
-------------------------------------------------------------------------
Euler rotation of hyperfine tensor to g-tensor
-------------------------------------------------------------------------
-------------------------------------------------------------------
Atom | Alpha Beta Gamma | Ax Ay Az
| [degrees] | [MHz]
-------------------------------------------------------------------
1H -90.1 42.7 89.9 -67.35 -25.41 -106.70
2H 90.0 17.4 -90.0 -67.36 -25.41 -106.71
3H -90.1 12.7 90.0 -67.36 -106.72 -25.42
-------------------------------------------------------------------
You can use the orca_euler software later if you want to rotate any of these for any specific application.
Comparison to experiment¶
To summarize our results, here is a table comparing the calculated and predicted data:
Property |
Exper. |
Theor. |
|---|---|---|
\(g\) |
2.0025 |
2.0026 |
\(A_{xx}\) |
-31.4 |
-25.4 |
\(A_{yy}\) |
-93.7 |
-106.7 |
\(A_{zz}\) |
-60.3 |
-67.3 |
\(A_{iso}\) |
-61.8 |
-66.5 |
With these, one can use tools such as EasySpin, which already has a specific function for reading orca outputs and simulate the EPR spectrum. The comparison between the theory prediction and an EPR spectra taken at 4K in a CH4 matrix is:
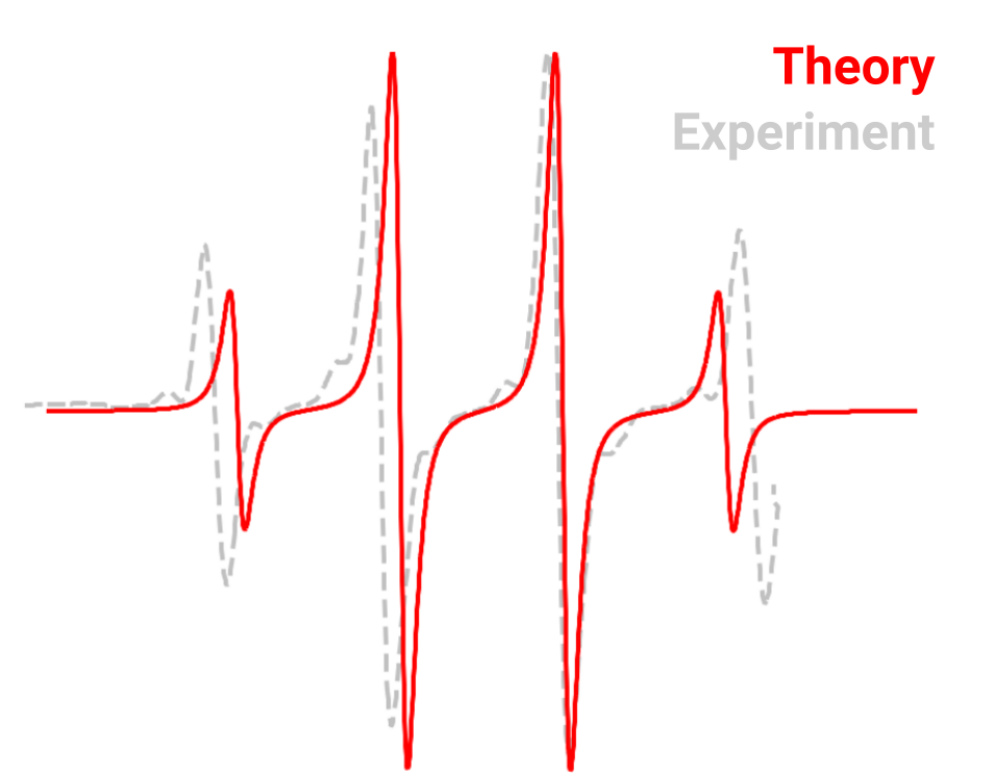
Of course differences here are expected due to impurities and matrices effects and the graphic was displaced to match the \(g\).
Important
The HFCs are quite sensitive to the geometry of the molecules! Be sure to have a good one before computing them.
Another example, the naphthalene anion radical¶
In aprotic solvents and in the presence of alkali metals, polyaromatics such as naphthalene can reduce to form radical anions [Garst1971], such as:
These will present nice EPR spectra, with two sets of four protons and thus more complex lines. To predict its spectrum, again one can start by optimizing the geometry of the anion:
!RI-B2PLYP DEF2-TZVP DEF2-TZVP/C TIGHTOPT
* XYZFILE -1 2 nap.xyz
We used a double-hybrid functional to add RI-MP2 correlation and get a better density and TIGHTOPT to obtain a good symmetrical geometry.
Now we run an EPR calculation, with the specialized EPR-III basis and RI-SOMF(1X) to speed-up the calculation of the integrals for the SOC part:
!RI-B2PLYP EPR-III AUTOAUX RI-SOMF(1X)
* XYZFILE -1 2 nap_optimized.xyz
%eprnmr
gtensor true
nuclei = all H {aiso, adip, aorb}
end
One can average the two sets of similar \(H_{\alpha}\) and \(H_{\beta}\) couplings (calculated from the MP2 relaxed density ) and use again some software like EasySpin to compute the predicted spectrum and compare it to the experiment. This will look like:
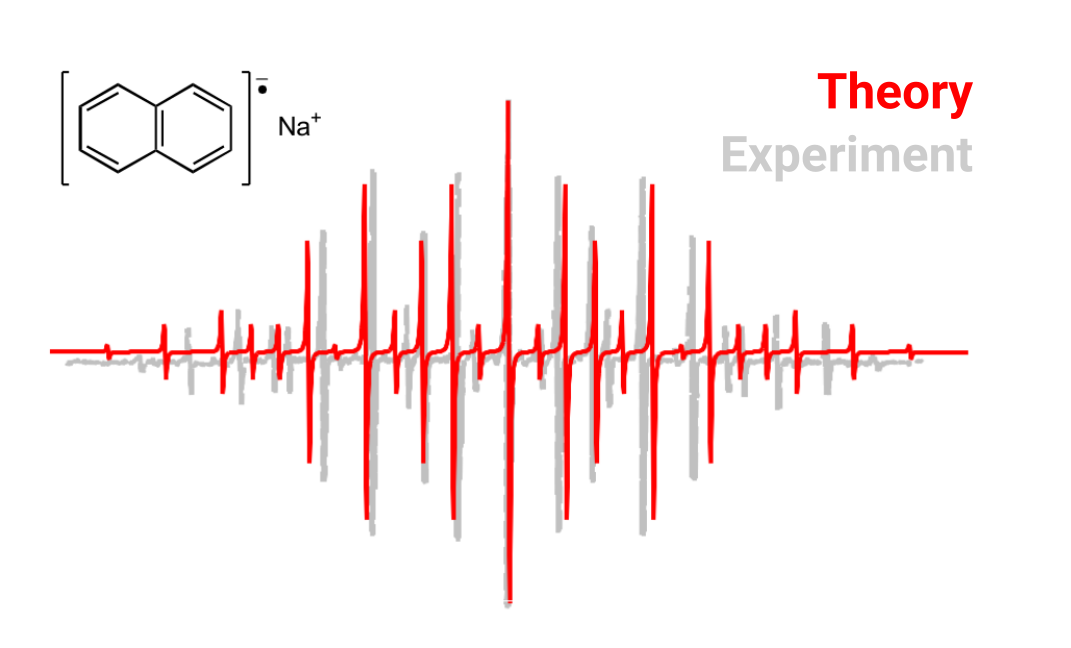
Visualizing the g tensor orientation¶
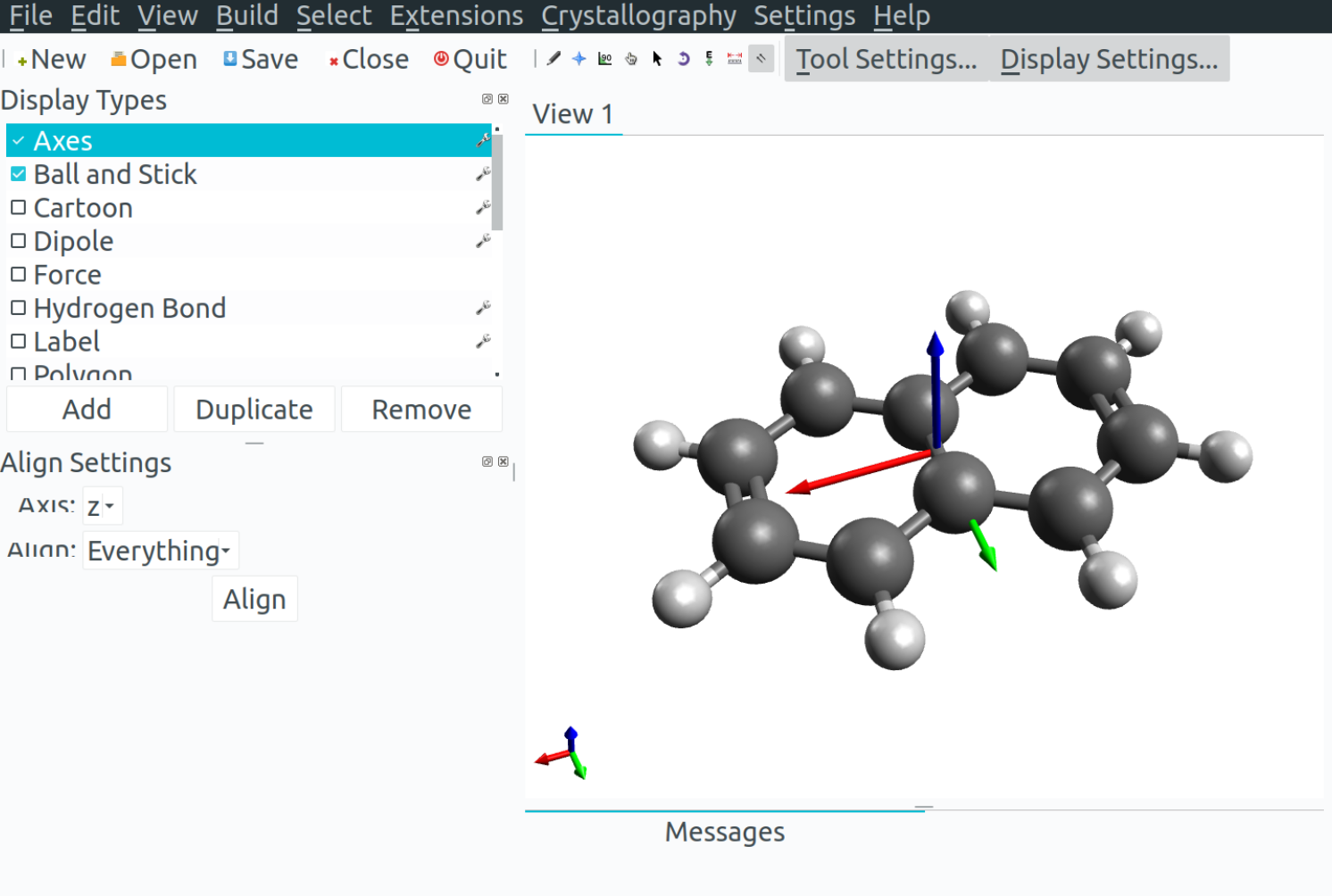
You can use Avogadro to visualize the orientation of the g tensor. Here is how it works: ORCA prints a basename.gori.xyz file, where dummy atoms are used to represent these vectors, He for the origin, Ne for the direction of \(g_z\), Ar for \(g_y\) and Kr for \(g_x\).
Open this file and select the "Align" option in Avogadro, on the right of the "E" used for optimization.
Now select the He-Ne bond and align the x axis and repeat in order for the y and z axis.
Delete the noble gas atoms.
Click on "Axes", the first option of the "Display Settings". Here it is!
Note
You can increase the size of the vectors by clicking on the "Axes" options. The order is red for x, green for y and blue for z (remember the RGB sequence).
Zero-field Splitting (ZFS)¶
If you have systems with \(S>1/2\), you might be interested in ZFS values. The ORCA_EPRNMR module can also be used to predict those. Let's try the example of cyclopentadienylidene carbene, which has known D value of \(0.408 cm^{-1}\) and a \(E/D\) ratio of \(0.046\) [Kirmse1971]. First, the geometry is optimized using:
!B3LYP DEF2-TZVP OPT
* XYZFILE 0 3 carb.xyz
to get the planar carbene:
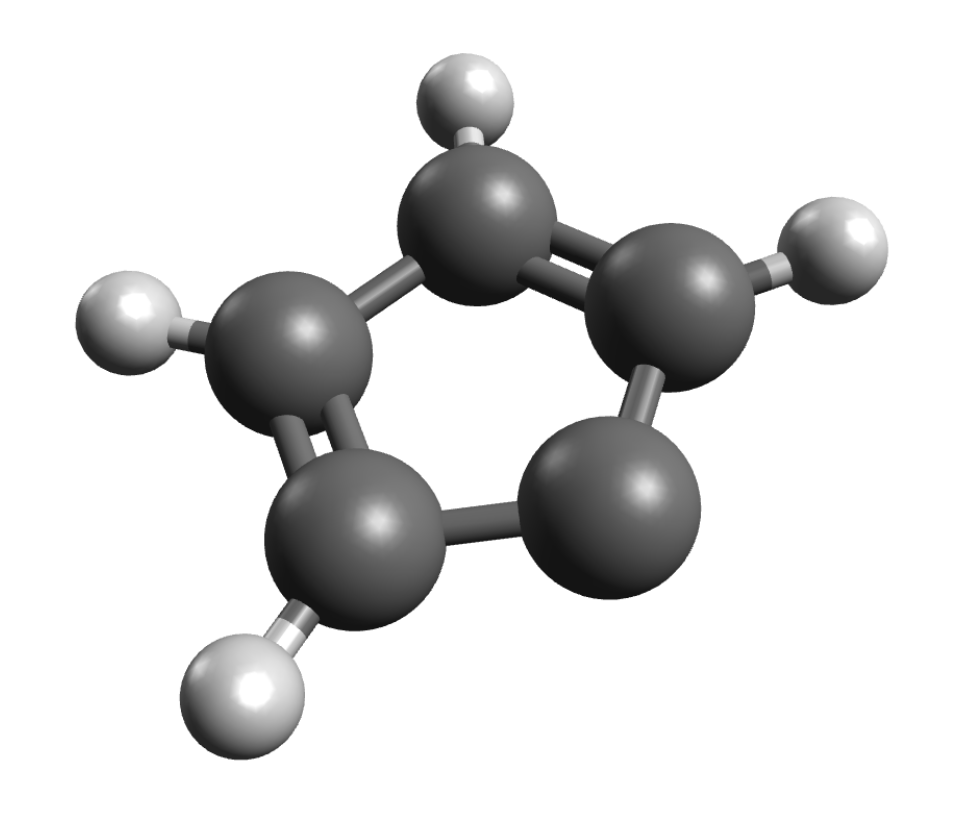
The ZFS can then be computed on simple DFT level using:
!B3LYP EPR-II AUTOAUX UNO
* XYZFILE 0 3 carb_optimized.xyz
%EPRNMR DTENSOR SSANDSO
DSS UNO
DSOC CP
END
The DTENSOR flag asks to include the spin-spin and the spin-orbit components of the \(D\) (SSANDSO), and the DSS and DSOC control the specific algorithms. For more information, consult the ORCA manual.
After the calculation of the SOC-related integrals, one should see the header:
------------------------------------------------------------------------------
ORCA EPR/NMR CALCULATION
------------------------------------------------------------------------------
GBWName ... ZFS.gbw
Electron density file ... ZFS.scfp
Spin density file ... ZFS.scfr
Spin-orbit integrals ... ZFS
Multiplicity ... 3
g-tensor ... 0
NMR chemical shifts ... 0
D-tensor ... spin-spin and spin-orbit
D(SS)-algorithm ... spin-density from UNOs
D(SOC)-algorithm ... CP (=Coupled-perturbed)
explaining the details of the calculation. The actual result is below the CP-SCF:
D = 0.425943 cm**-1
E/D = 0.015874
which is again close to the experiment, considering that we are using DFT and a single-reference method.
Important
This was intended to be a simple demonstration. In general, much better results for ZFS can be obtained from multi-reference theories such as CASSCF or MRCI. Nonetheless, it is still quite good and is easily applicable to large systems.
Starting structures¶
Napthalene
C 2.41963 0.74477 0.00000
C 2.44765 -0.64677 -0.00000
C 1.25295 -1.36872 0.00000
C 0.01423 -0.70692 0.00000
C -0.01423 0.70692 -0.00000
C 1.19684 1.41804 -0.00000
C -1.25295 1.36871 0.00000
C -2.44765 0.64677 -0.00000
C -2.41963 -0.74477 0.00000
C -1.19684 -1.41804 -0.00000
H 3.34913 1.30792 -0.00000
H 3.39906 -1.17205 0.00000
H 1.29505 -2.45547 -0.00000
H 1.19516 2.50560 0.00000
H -1.29505 2.45547 -0.00000
H -3.39906 1.17205 0.00000
H -3.34913 -1.30792 -0.00000
H -1.19516 -2.50561 0.00000
Cyclopentadienylidene carbene
C -1.73238 3.09526 0.00000
C -2.43358 1.94496 -0.00000
C -0.30953 2.77063 0.00000
C -0.17373 1.43249 -0.00000
C -1.48252 0.90001 -0.00000
H -2.13310 4.09634 0.00000
H -3.50614 1.82921 -0.00000
H 0.48447 3.50033 0.00000
H 0.74363 0.86480 -0.00000